100 Nano-Stories: Mie Scattering & Mie Theory!
Episode #77: Concepts, Properties, and The Sky!
Preface! ✨
It’s your favorite material science & nanotechnology enthusiast! Today, we will cover a new theory in the series of nanotechnology and aerogel: Mie Theory!
If you need a reminder on one main application of the Mie theory in aerogels, check out this article below!
Don’t worry, it’s only a 4-minute read! 😁
TL;DR → Beer’s Law Violations (Part 3)! 🔑
- The incoming wavelengths of light that enter the aerogel are not one wavelength of light only (monochromatic).
- Mie Scattering → A form of scattering that can occur in a spherical particle that either has the same diameter or larger than a certain wavelength of light that will enter the molecule/spherical particle (aka incident light).
- Because there is more than one wavelength of light (incident light) that passes through the aerogel, it breaks one of the laws from Beer’s Law, and since some of the incident light is bigger than the particles, it will follow Mie Scattering!
Now that we have some of the main concepts down, let’s introduce everything you must know about Mie Scattering to apply the concepts to transparent aerogels!
Mie Scattering + Theory! 💡
Concepts! 🔑
If you remember from old articles on Rayleigh Scattering, we can find that scattering in the sky. The same thing happens with Mie Scattering, except Mie Scattering doesn’t depend on the wavelength of the light. Mie Scattering is what makes the sun look so bright and white from the ground.
The reason why Mie Scattering makes this white light appear around the Sun because the incident light carries more intensity as it moves forward, and creates less saturation in the sky.
Saturation → A color with no mixture of white (pure red, pure purple, etc.).
If confused, here is a photo comparing Rayleigh Scattering & Mie Scattering:
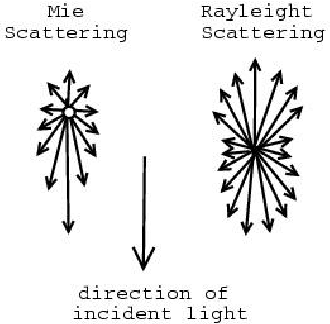
Properties of Mie Scattering! 🔑
In Rayleigh Scattering, the light will prefer particle sizes/molecules that are shorter than a specific wavelength of light. Rayleigh Scattering prefers shorter wavelengths of light, such as blue. This specific wavelength of light is bounded by the equation below (1/λ⁴):
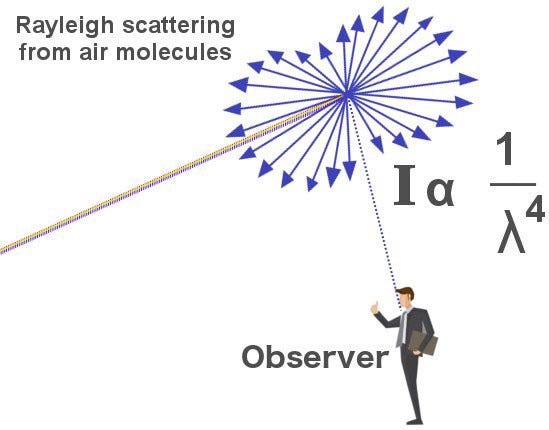
But Mie Scattering will dominate Rayleigh Scattering if the particle/molecules are larger than the specific wavelength that dominates Rayleigh Scattering. In other words, Mie Scattering will happen if a particle/molecule has about a radius of greater than 2 micrometers or more because the particle is much bigger than any wavelength of light (visible, some infrared light)!

Closing Thoughts! 💭
Now we know what the effects are of Mie Scattering are and how it affects the world we live in (with a bit of help from Rayleigh Scattering 😉)!
See you tomorrow to talk more about the Frontal Lobe and mathematics of Mie Scattering + Theory! ✌🏽
Vocabulary! 📓
Mie Scattering → A form of scattering that can occur in a spherical particle that either has the same diameter or larger than a certain wavelength of light that will enter the molecule/spherical particle (aka incident light).
Monochromatic → Contains only one wavelength of light and/or one color.
Incident Light → The light that will enter the molecule/spherical particle/material.
Wavelength Symbol → λ
Rayleigh Scattering → The wavelength of a certain ray of light is short, then the wavelength of light will scatter more than a ray of light where the wavelength is larger, and Rayleigh Scattering tends to scatter blue light because of its wavelength (450–485 nanometers).
Bonus Resources! 📓
Previous “100 Nano-Stories!” 🔖
© 2021 by Carlos Manuel Jarquin Sanchez. All Rights Reserved.
